Stem Cell Therapy Plans: The Definitive 2026 Editorial Reference
The field of regenerative medicine has reached a pivotal juncture where the theoretical potential of cellular biology is manifesting as a structured clinical reality. For decades, the discourse surrounding cellular therapeutics was relegated to the fringes of experimental science or the hyperbole of speculative marketing. However, as we move through 2026, the landscape has matured into a disciplined medical sector characterized by rigorous manufacturing standards, precise delivery mechanisms, and a deeper understanding of “Signal Modulation.” The challenge for the contemporary patient or medical strategist is no longer finding access to these treatments, but rather navigating the significant variability in quality, ethics, and biological viability.
A premier regenerative strategy is defined by its recognition that cells are not “pills,” but dynamic biological agents that interact with a host’s unique systemic environment. The shift from “one-size-fits-all” injections to highly personalized regenerative roadmaps requires an analytical understanding of the “Niche Environment,” the specific micro-anatomical conditions of the target tissue. Identifying a path toward meaningful recovery involves a synthesis of immunology, molecular signaling, and long-term metabolic governance.
Understanding “stem cell therapy plans.”
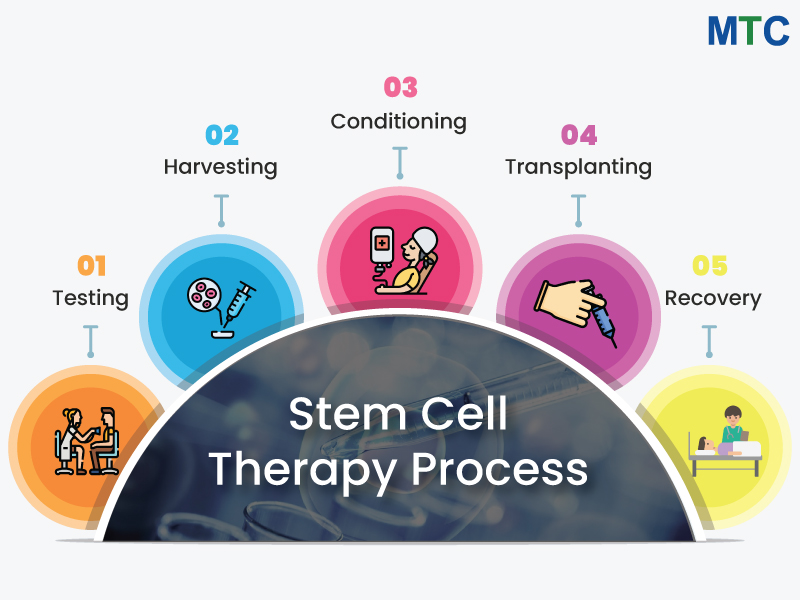
To engage with stem cell therapy plans effectively, one must first decouple the “biological material” from the “clinical strategy.” In a professional regenerative context, a plan is a longitudinal protocol. It is not a single injection event but a multi-phase biological intervention that includes pre-treatment priming, targeted delivery, and post-procedural signaling support. A plan might utilize the highest quality mesenchymal cells, but if the recipient’s tissue is in a state of chronic oxidative stress, the cells will likely enter senescence or be cleared by the immune system before they can exert a paracrine effect.
Multi-Perspective Explanation
From a Molecular Perspective, these plans are judged by their “Secretome Profile”—the specific cytokines and growth factors the cells release to stimulate local repair. From a Structural Perspective, evaluation is based on the “Scaffolding Integration,” or how well the cells adhere to the damaged tissue, such as a tendon or joint capsule. Finally, from a Regulatory Perspective, a plan must be scrutinized for its compliance with “Current Good Manufacturing Practice” (cGMP) standards, ensuring that the cellular “product” is free from contaminants and possesses verified potency.
Oversimplification Risks
The most significant risk in cellular planning is “Dose-Dependency Fallacy”—the belief that injecting more cells always leads to a better result. In reality, an excessive cellular load in a confined space can lead to localized hypoxia (lack of oxygen) and cell death. A professional assessment avoids these oversimplifications by focusing on “Precision Concentration” and the timing of the “Inflammatory Window,” ensuring the cells are introduced when the environment is most receptive to signaling.
Contextual Background: The Evolution of Regenerative Medicine
The trajectory of cellular therapy has moved from the early bone marrow transplants of the 1950s,s primarily used for hematological cancersto the “Paracrine Revolution” of the current era. Early regenerative attempts were built on the “Replacement Model,” the idea that injected stem cells would simply transform into new bone, cartilage, or heart muscle.
Conceptual Frameworks and Mental Models for Evaluation
Experienced clinicians utilize specific mental models to evaluate the viability of a regenerative plan.
1. The Seed-and-Soil Framework
This model posits that the cells are the “Seed” and the patient’s tissue is the “Soil.” A plan that focuses only on the cells while ignoring the soil (the patient’s systemic inflammation, nutrition, and blood flow) is fundamentally flawed. The best plans prioritize “Soil Preparation”—often through diet, specific supplements, or localized shockwave therapy—weeks before the cells are introduced.
2. The Paracrine Signaling Mental Model
This shifts the focus from “Cell Survival” to “Signal Duration.” Since many injected cells are cleared by the body within weeks, the goal of a high-tier plan is to trigger a “Healing Cascade” that continues long after the exogenous cells are gone. This model evaluates the plan based on its ability to flip the tissue’s internal switch from “Degenerative” to “Regenerative.”
3. The “Homing” Logic
Stem cells have a natural affinity for inflammation. The Homing model helps a clinician decide between systemic (IV) and localized (injection) delivery. If the target is a specific knee joint, localized delivery is preferred; if the target is systemic autoimmune regulation, the plan should leverage the cells’ ability to navigate through the vascular system to multiple sites of injury.
Key Categories and Physiological Variations
The regenerative landscape is categorized into distinct “Operational Profiles,” based on the source of the cells and the mechanical intent of the plan.
| Profile | Source | Primary Benefit | Significant Constraint |
| Autologous (BMAC) | Patient’s Bone Marrow | Zero risk of rejection; FDA compliant. | Potential for low cell count in older patients. |
| Autologous (Adipose) | Patient’s Fat Tissue | Very high cell yield; easier harvest. | Requires enzymatic digestion (regulatory hurdles). |
| Allogeneic (Umbilical) | Donor Cord Tissue | “Young” cells; high expansion potential. | Requires strict donor screeninganda cold chain. |
| Exosome Therapy | Cellular Byproducts | Acellular (no DNA); low inflammation risk. | Short half-life; requires frequent dosing. |
| VSELs | Very Small Embryonic-Like | Potential for multi-tissue differentiation. | Highly experimental; difficult to isolate. |
Realistic Decision Logic
The selection of a profile must be driven by the Patient’s “Biological Age.” For a 25-year-old athlete with an acute injury, an autologous (from self) plan is often superior because their own cells are still robust and active. For a 75-year-old with multi-joint osteoarthritis, an allogeneic (donor) plan utilizing “younger” umbilical cord-derived MSCs may be necessary, as their own “cellular pharmacy” has been depleted by decades of aging.
Detailed Real-World Scenarios and Decision Logic

The “Chronic Tendinopathy” Case
A 50-year-old with a persistent Achilles tendon tear that has failed physical therapy.
-
Decision Point: PRP (Platelet-Rich Plasma) vs. MSC (Stem Cell) Therapy.
-
Analysis: PRP provides growth factors but no “living” signaling platform. The “Soil” is avascular (poor blood flow), meaning the PRP will wash out quickly.
-
Outcome: The plan utilizes MSCs embedded in a “Fibrin Scaffold” to hold the cells in place, providing months of sustained signaling to the tendon.
The “Neuro-Regenerative” Attempt
A patient seeking support for early-stage neurodegeneration.
-
Constraint: The Blood-Brain Barrier (BBB) limits cell entry.
-
Decision Point: IV Delivery vs. Intrathecal (Spinal) Delivery.
-
Second-Order Effect: While IV is easier, the “Homing” logic suggests that intrathecal delivery bypasses the systemic filters (lungs/liver), allowing a higher concentration of “Signal” to reach the central nervous system.
Planning, Cost, and Resource Dynamics
The financial dynamics of cellular medicine are defined by “Expansion Costs” and “Regulatory Overhead.”
Range-Based Operational Cost Table (US & International 2026)
| Category | Typical Session Fee | Plan Duration | Total Budget Estimate |
| Localized Orthopedic | $3,000 – $7,000 | 1 – 2 Sessions | $5,000 – $12,000 |
| Systemic/IV Wellness | $5,000 – $15,000 | Annual Cycles | $10,000 – $30,000 |
| Advanced Neurological | $15,000 – $40,000 | Multi-Phase | $25,000 – $60,000 |
| Exosome Protocols | $2,000 – $5,000 | 3 – 5 Sessions | $8,000 – $15,000 |
Note: In 2026, the “best” value is found in plans that include “Pre-habilitation. A professional plan invests $1,000 in metabolic optimization before spending $9,000 on the cells.
Support Systems, Tools, and Strategic Resources
A successful cellular reconstruction relies on a “Regenerative Stack” of specialized resources:
-
Flow Cytometry: The “Quality Control” tool used to verify that the cells being injected are actually the type and quantity promised.
-
Image-Guided Delivery (Ultrasound/Fluoroscopy): Essential to ensure cells are placed within the specific “Niche” (e.g., the intra-articular space of the hip).
-
Cold-Chain Logistics: A governance system ensuring cells are stored at -190°C and thawed only minutes before injection.
-
Cytokine Testing: Blood panels that measure IL-6 and TNF-alpha to track the systemic “Inflammatory Baseline.”
-
Peptide Synergies: The use of BPC-157 or Thymosin Beta-4 to “prime” the tissue and enhance the stem cells’ homing signals.
-
Hyperbaric Oxygen (HBOT): Often used post-procedure to increase localized oxygen, supporting the survival of the newly introduced cells.
Risk Landscape and Failure Modes
Even the most prestigious cellular plans harbor compounding risks.
-
The “Lung Trap”: When cells are delivered via IV, a large percentage are caught in the fine capillaries of the lungs (the pulmonary first-pass effect), reducing the dose that reaches the target organ.
-
Ectopic Tissue Formation: A rare failure where stem cells differentiate into the “wrong” type of tissue (e.g., bone forming in a muscle), usually caused by poor signal management.
-
Immune Rejection (Allogeneic): While MSCs are “immune-privileged,” the body may still recognize and destroy donor cells if they are not processed correctly.
-
Regulatory Volatility: A plan may be disrupted by sudden changes in FDA or local health authority enforcement, leading to the “Orphaning” of a patient mid-protocol.
Governance, Maintenance, and Long-Term Adaptation
To maintain the gains of a regenerative intervention, patients must adopt a “Governance” mindset.
-
The “Metabolic Service Plan”: Since stem cells respond to the host environment, maintaining low insulin levels and high nutrient density is the “Service Plan” for cellular longevity.
-
Review Cycles: A plan should be evaluated every 6 months using functional imaging (MRI) or biochemical markers to determine if a “Booster” or exosome cycle is needed.
-
Adaptation Triggers: If a patient experiences a new systemic trauma (such as a severe viral infection), the regenerative plan must pivot to address the new inflammatory load before returning to the primary focus.
Measurement, Tracking, and Evaluation Signals
How do you measure the success of a cellular therapy plan?
-
Leading Indicators: Reduced reliance on NSAIDs; improved sleep quality (as systemic inflammation drops); increased range of motion within 30 days.
-
Qualitative Signals: The “Return to Function”—the ability to perform activities of daily living without the “grinding” or “heaviness” associated with degeneration.
-
Documentation: Maintaining a “Cellular Passport” that includes the Certificate of Analysis (CoA) for the cells used, including viability percentages and endotoxin levels.
Common Misconceptions and Oversimplifications
-
“Stem Cells Cure Everything”: They are “Biological Modulators.” They help the body fix itself; they do not perform “magic” on dead tissue.
-
“Vatican/Overseas Clinics are Always Better”: Some are world-class, but many leverage a lack of regulation to sell “Stem Cell Tourism” with no verified cell counts.
-
“Amniotic Fluid is Stem Cell Therapy”: Most commercial amniotic products are “Acellular”—they contain growth factors but no living stem cells.
-
“One Injection is Enough”: Chronic degeneration usually requires a “Loading Phase” of 2-3 sessions followed by annual maintenance.
-
“Stem Cells Cause Cancer”: While a theoretical risk with embryonic cells, there is no clinical evidence that adult Mesenchymal Stem Cells (MSCs) cause tumors in humans.
-
“The Younger the Cells, the Better”: While often true, the “Compatibility” of the cell’s signaling profile with the patient’s specific disease state is more important than age alone.
Ethical and Practical Considerations
The ethics of cellular medicine in 2026 revolve around “Informed Consent” and “Claims Integrity.” A plan that promises a “Cure” for incurable diseases is ethically compromised. Intellectual honesty requires acknowledging that regenerative medicine is a high-reward but high-variability field.
Conclusion
The architecture of biological renewal is a strategic exercise in aligning cellular precision with systemic health. It is a transition from a model of “Managing Decay” to a model of “Fostering Resilience.” Whether you are considering a localized joint injection or a systemic wellness protocol, success depends on the alignment of technical precision, anatomical respect, and lifelong maintenance. In 2026, the ultimate metric of a successful cellular plan is not the “Magic” of the injection, but the stability of the recovery, the assurance that the biological signaling has permanently shifted the body toward a state of self-directed repair.